Aerogen Pro
(formerly Aeroneb Pro)
The manufacturer Aerogen Ltd. has decided to replace the established brand name Aeroneb with the company name Aerogen. Technically the products have not been changed.
Eefficient
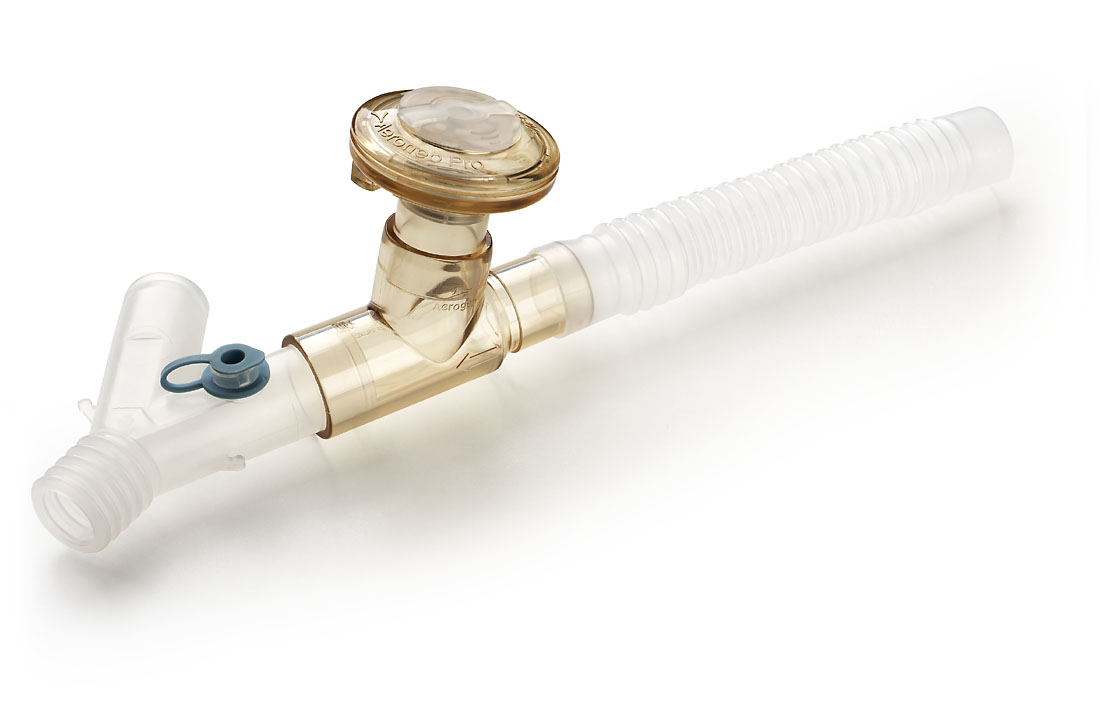
Saves time and moneyAerogen Pro control module (formerly Aeroneb) connected to the Pro nebuliser and autoclavable adult T-piece
Maximum therapeutic success through optimized particle spectrum. Easy handling and universal application possibilities - from intensive care to home use
Flexible
The device works as a hand-held device or in a tube system from neonatal to adult without influencing the ventilation parameters, even during inhalation or refilling The system can be operated with mains or rechargeable battery (via integrated NiMH battery).
Approved as hand nebulizer, as well as for all common homecare, transport and intensive care ventilation systems (incl. high frequency), it reliably nebulizes all nebulisable medications, suspensions, steroids.
Effective and economical, ecological
Aerogen Pro nebulizes a 6ml fill to the last drop within 15 minutes. This happens silently and without heating or denaturation. This reduces the costs for nebulization and medication.
The nebulizer unit can be used by multiple patients and is sterilizable and autoclavable. This system does not require any consumables and thus additionally protects the environment.
The Aerogen® Pro Nebulizer System
An extraordinary medication nebuliser system that saves time and money and can be used flexibly in the complete hospital operation - in the practice - in the homecare sector and on the road.
The Aerogen Pro offers effective administration of inhaled medication both on the ventilator (intensive care and home ventilation) and as a hand nebuliser for on-the-go use for treatment from newborn to adult. Using the unique Aerogen aerosol generator, the Aerogen Pro produces a slow fine particle aerosol without propellants, inefficient compressors or expensive ultrasound technology. The Aerogen Pro solves your problem of poor drug delivery during ventilation and places up to four times more drug through an ET tube (in vitro) than small volume disposable nebulizers (SVNs). These SVNs have been shown to place only between 1-3% of the drug used through the ET-tube in the lungs and only 2-18% in non-intubated patients.
 | PRODUCT SPECIFICATIONS
Physical:
Weight nebuliser: 25 grams
Weight control module: 230 grams
capacity nebuliser beaker: 10 ml
Electric:
Power consumption:< 6.5 Watt (charging current)
< 2.0 Watt Nebulisation
Power supply (input 100 to 240 VAC 50/60Hz, output 9 V) or built-in battery (4.8 V nominal output)
Patient protection insulation: Tested according to IEC 60601-1 and AAMI ES1
Performance:
See performance table
Noise generation: < 20 dBA
Frequency nebulizer: 128 kilohertz (kHz)
|
Documents for download:
Operating instructions (german)
Product leaflet (german)
Article list (german)
Detailed view